[[[SUMMARY_START]]]
The global wellness economy has continued to grow, with new data putting it at about $6.8 trillion in 2024 and projecting further gains through 2029.
At the same time, regulators in the US and Europe are moving more aggressively against misleading health claims and gray-market “wellness” products.
Recent actions involving compounded GLP-1 weight-loss drugs, influencer marketing, and botanical supplement claims highlight how hard it can be for consumers to judge what is proven and what is promotional.
The result is a booming marketplace where the boundaries between medicine, supplements, and lifestyle services are still often unclear.
[[[SUMMARY_END]]]
Wellness is getting bigger, richer, and more mainstream. Clear standards for what works, what is safe, and what can be claimed in advertising are not keeping pace.
Fresh industry estimates show continued rapid expansion across many wellness categories, from beauty and nutrition to mental health services and “wellness real estate.” But recent regulatory moves in the United States and Europe underscore a different trend: authorities are spending more time on misleading marketing, unapproved products, and health claims that look like medicine but are sold as lifestyle upgrades.
That growth is visible across everyday consumer life. More employers are buying workplace wellness services. More travelers are seeking wellness-focused hotels. More people are spending on supplements, beauty, and weight management.
But the same broad definition that helps wellness scale also makes it hard to police. “Wellness” can mean gym memberships and sleep coaching, but it can also mean injections, pills, and medical-sounding claims that cross into regulated territory.
## A bigger market with fuzzy boundaries
Wellness is not a single industry with one rulebook. It is a collection of sectors that sit across consumer goods, healthcare, hospitality, and digital services.
That creates room for fast innovation. It also creates room for confusion.
Many products and services are sold with scientific language that is difficult for non-experts to evaluate. Marketing often blends nutrition advice with disease-related claims. Social media adds speed and scale. Influencers can promote “protocols” that look clinical, while disclaimers and fine print may be easy to miss.
This is not just a communication problem. It can become a safety issue when consumers are pushed toward unapproved substances, or away from proven care.
## US focus turns to compounded GLP-1 drugs and mass marketing
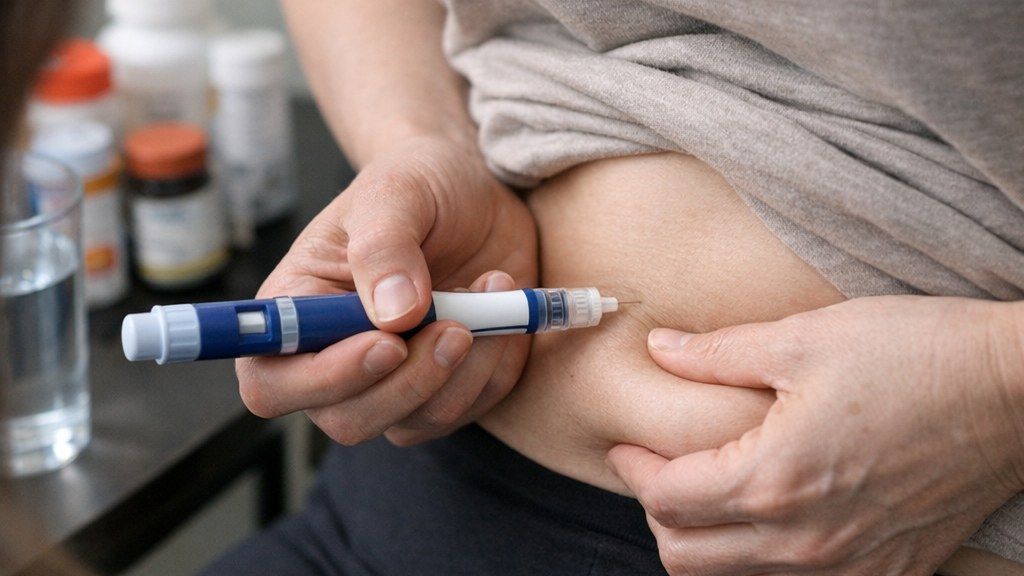
In the United States, one of the sharpest recent examples involves GLP-1 drugs used for diabetes and weight loss.
As demand surged, many telehealth businesses and compounding pharmacies offered compounded versions of GLP-1 medicines. Compounded drugs are not the same as FDA-approved products. Compounding can be appropriate in limited, patient-specific situations, but regulators have warned against mass marketing of compounded versions as easy substitutes for approved drugs.
In late 2025, the FDA said it sent warning letters related to compounded GLP-1 products and marketing. In February 2026, the agency said it intended to take action to restrict GLP-1 active pharmaceutical ingredients used in non-FDA-approved compounded drugs that are being mass-marketed as alternatives to FDA-approved medicines.
The debate has also spilled into corporate and legal battles. In early February 2026, the telehealth company Hims & Hers said it would launch a cheaper, off-brand version of a weight-loss pill tied to semaglutide, and Novo Nordisk said it would sue, describing the product as unapproved and untested.
Separately, concerns about unproven “peptides” marketed for muscle, skin, and longevity have also drawn public attention. Some of these substances are promoted online with limited or no human evidence, adding another layer to the broader “biohacking” and wellness market.
## Europe reinforces limits on health claims in advertising
In Europe, enforcement is also tightening around what sellers can claim.
In April 2025, the EU’s top court ruled that advertising with unauthorized health claims for herbal food supplements is prohibited while those claims are still under assessment. The decision raised the bar for companies that have relied on “traditional use” narratives or emerging research to imply medical benefits.
In the United Kingdom, the advertising regulator has also acted against supplement promotions that suggested products could treat or cure conditions. In a 2025 ruling covered by a medical journal, an influencer was found to have breached advertising rules by promoting a supplement with claims connected to ADHD and by encouraging people to stop prescribed medication.
The UK regulator has also upheld complaints against multiple advertisers for making medicinal claims in paid ads for products that are not authorized medicines or medical devices.
## What “clarity” would look like for consumers
The common thread is not that wellness is inherently misleading. It is that the category often mixes three very different worlds:
- Evidence-based medical treatment, which requires approvals and clear labeling.
- Consumer products like foods and supplements, which have different regulatory pathways.
- Lifestyle services, where outcomes are often subjective and hard to measure.
When these worlds blend in one ad, one influencer post, or one telehealth checkout flow, it becomes harder for consumers to know what standard of proof is being used.
Regulators are signaling that disclaimers are not enough if the overall message implies disease treatment or if the science is overstated. The next phase of wellness growth is likely to depend not only on demand, but also on clearer lines between marketing and medicine.
AI Perspective
Wellness is growing because many people want more control over sleep, stress, weight, and daily health. The challenge is that a large, fast-moving market can reward bold claims even when the evidence is limited. Clearer labeling and stricter rules on medical-style promises can make it easier for consumers to choose what is both safe and effective.































































-b9c5792e.jpeg)













-dce9c62e.jpeg)